The Bite That Held the Color
For millennia, a single chemistry step separated textile dye that lasted centuries from dye that washed out in a season.
The Botanical Mordant Calculator brings those precise ratios to your browser — calibrated to centuries of guild practice.
✦ Transparency note: This article was written by AI and reviewed by the author. All factual claims were independently verified (with another prompt) before publication. Mistakes may still happen.
Disclaimer: The information in this post is for educational and informational purposes only. It does not constitute financial, legal, or professional advice. The author is not liable for any financial loss or damages arising from use of this information. Data, pricing, and availability referenced here may be out of date — always verify independently before acting on it.
The Word Says It All 🧪
The word “mordant” comes from the Latin mordere — to bite.
That is not a metaphor. In the chemistry of natural dyeing, the mordant literally bites into the fiber before the dye ever touches it. It forms a metallic coordination complex with the cellulose — a tight chemical grip — and then the dye molecule bonds to that complex. Without the bite, the dye sits on top of the fiber with nothing to hold it there. One wash, and it is gone.
This is why some ancient textiles still carry their color today, thousands of years after they were made. Not because the dye was extraordinary. Because someone knew to mordant first. 😮
The Most Strategic Salt in Medieval Europe ⚗️
For over a thousand years, the most important mordant in the world was alum — potassium aluminum sulfate — a mineral salt mined from natural deposits. And for centuries, much of the European supply came from deposits along the Anatolian coast, near the ancient city of Phocaea.
European textile guilds — in Flanders, Florence, and England — depended on that alum to fix their dyes. Red from madder (Rubia tinctorum), yellow from weld (Reseda luteola), blue from woad: none of them would hold on wool or linen without a mordant bath first. The trade was lucrative, and the leverage it gave suppliers over the great cloth-producing cities was real.
Then in 1462, substantial alum deposits were discovered at Tolfa, in the hills northwest of Rome. The discovery transferred the supply chain — and the economics of European textile color — almost overnight. 🎯
The chemistry, however, stayed exactly the same: alum, fiber, dye, in the right proportions, in the right sequence. That ratio — how much alum per gram of dry fiber — is precisely what the Botanical Mordant Calculator encodes today.
What the Bite Actually Does 🔬
Here is the chemistry, as plainly as possible.
Cellulose fibers — cotton, linen, hemp, ramie — are notoriously difficult to dye. Unlike wool and silk (which are protein fibers with plenty of chemical attachment points), plant-based fibers are chemically inert. Natural plant dyes do not have strong enough affinity to bond to cellulose on their own.
Mordants solve this by providing an intermediate layer. Potassium alum dissolves in water and dissociates into aluminum ions. Those ions form coordination bonds with the hydroxyl groups on the cellulose fiber. The dye molecule then bonds to the aluminum center. The result is a dye–mordant–fiber structure that withstands washing, light exposure, and time — if the ratio is right.
Iron mordanting (ferrous sulfate) works by the same mechanism but “saddens” the hue — shifting yellows toward olive, golds toward grey-green. Dyers use iron deliberately for tonal shifts, not just as a convenience. A madder red with no iron is coral; with iron, it shifts toward terracotta and then brown.
Tannin is different again: it functions as a pre-mordant, applied before the alum bath to increase the fiber’s surface attachment points. Cellulose fibers benefit significantly from this preparatory step. The tool includes tannin as a third option for exactly this use case. 🧪
If the iron chemistry angle interests you, it also drives the cyanotype process — a completely different application of the same principle. That story is in The Blueprint Was Always a Photograph.
The WOF System: Why 20% Is Not Arbitrary 📐
The key number in mordanting is the WOF — Weight of Fiber. Mordant quantity is expressed as a percentage of the dry fiber weight.
20% WOF alum = 20 g of alum per 100 g of dry fiber.
This ratio is scale-independent. It works identically for 50 g of cotton thread in a kitchen pot and 5 kg of hemp cloth in a commercial vat. The percentage holds.
The WOF system was refined through generations of guild practice and documented in 19th-century textile literature. The ratios were not invented by any single author — they were empirically derived by dyers over centuries, then collected and printed so that anyone with a kitchen scale and a mordant supplier could reproduce them consistently.
The tool applies these same ratios with one important addition: it calculates water bath volume as a second output. The recommended minimum is 30:1 water-to-fiber by weight; 40:1 for iron, where even distribution matters more. This is the number that most informal dyeing guides skip — and it is the one responsible for uneven mordanting when ignored. 💡
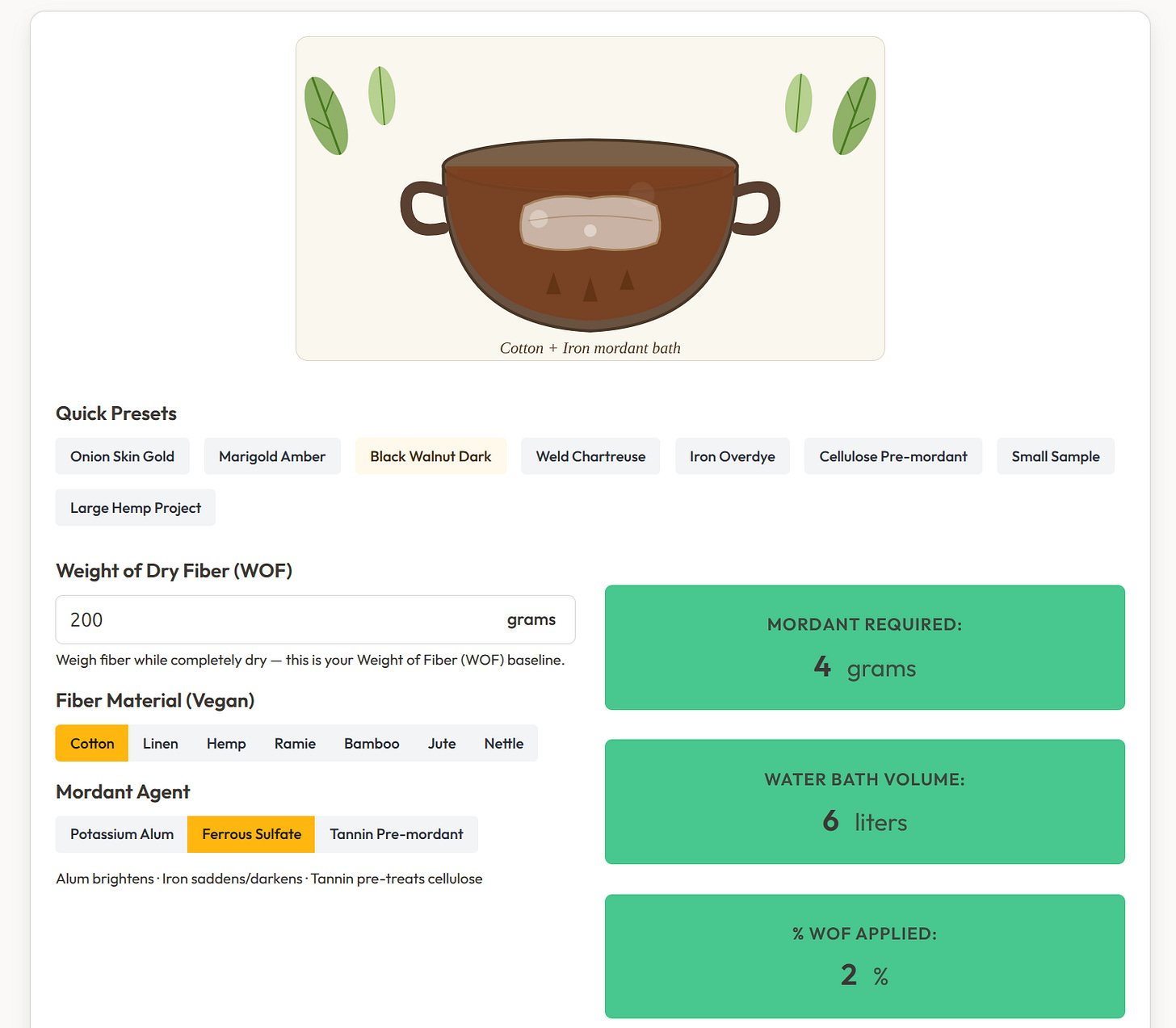
What the Tool Does 🛠️
The Botanical Mordant Calculator takes three inputs:
Weight of dry fiber (grams)
Fiber material — Cotton, Linen, Hemp, Ramie, Bamboo, Jute, or Nettle
Mordant agent — Potassium Alum, Ferrous Sulfate, or Tannin Pre-mordant
And returns four outputs:
Mordant required (grams)
Water bath volume (liters)
% WOF applied
Color effect — a description of what that mordant does to the final hue
The defaults are sensible: 150 g cotton + alum returns 30 g alum, 4.5 L water, 20% WOF, and the note that alum produces “brightened, clarified colors — ideal for vivid yellows, golds, and reds.” 🎯
Below the calculator, the tool includes a dye plant reference table — ten proven plant-mordant pairings with expected color results and notes on technique. Onion skins, marigold, weld, black walnut hulls, madder, chamomile, St John’s Wort, woad, sumac, and nettle, each with the recommended mordant and any special handling notes.
Eight Presets, Eight Starting Points 🗺️
The tool includes eight quick-load presets. Here is what each one is actually for:
Onion Skin Gold — the beginner’s entry point; onion skins with alum produce a high-yield, vivid gold on cotton; very forgiving, highly recommended as a first attempt
Marigold Amber — alum mordant for warm yellow-gold; the dye plant note recommends 100–200% WOF of flowers for strong color saturation
Black Walnut Dark — no mordant needed; black walnut hulls are self-mordanting; the iron preset deepens this toward near-black
Weld Chartreuse — the tool’s own note calls weld “the best lightfast yellow available to natural dyers”; alum mordant, reliable and historically important
Iron Overdye — ferrous sulfate to sadden and shift hue; useful for overdyeing existing color toward olive and grey-green tones
Cellulose Pre-mordant — tannin only, no metal mordant; the preparatory step for difficult cellulose fibers before a subsequent alum bath
Small Sample — scaled for test skeins and sampling runs before committing to a full batch
Large Hemp Project — scaled for large cellulose fiber batches where water bath volume matters most
The Onion Skin Gold preset is where to start. Onion skins are abundant, free, and produce a remarkably stable and vivid gold on alum-mordanted cotton. The WOF is generous, the process is forgiving, and the result teaches you what correctly mordanted fiber looks and feels like before you ever introduce a dye bath. 😊
How to Use It
Load the Onion Skin Gold preset, or enter your fiber weight manually
Select your fiber type and mordant agent
Read: mordant grams + water bath volume
Weigh your mordant on a kitchen scale; dissolve in hot water
Submerge the wet fiber (pre-wet it before the bath), bring to a gentle simmer — not a boil
Hold 45–60 minutes, cool in the bath, rinse gently, move directly to the dye bath while still wet
One note the tool flags explicitly: always weigh fiber dry. Wet fiber is heavier, and weighing after wetting will throw off the WOF calculation.
For supplies — alum mordant, dried dye plants, natural fiber:
→ Potassium alum mordant on Amazon | → Natural dyeing books on Amazon
Affiliate disclosure: This post contains Amazon affiliate links. I may earn a small commission at no extra cost to you.
The Color That Synthetic Dyes Almost Erased 🎨
In 1856, an 18-year-old chemistry student named William Henry Perkin was trying to synthesize quinine in a home laboratory on Cable Street, East London. He failed, but the residue in his flask was a vivid purple — mauveine, the first commercially mass-produced synthetic dye, which he brought to market under names including “aniline purple” and “mauve.”
That was the opening shot. The decisive blow to natural dyeing came thirteen years later, when chemists Carl Graebe and Carl Liebermann synthesised alizarin — the active colouring compound in madder root — in 1869. Industrial production followed almost immediately. France’s madder-growing regions, which had supplied a significant share of the world’s red dye, peaked in 1881 and had effectively collapsed by 1886. A crop that had dyed the red coats of European fashion for centuries became commercially worthless within five years. Synthetic dyes were cheaper, more consistent, and required no mordanting at all.
But here is what synthetic dyes lost in the process: botanical complexity. Plant dyes produce colors that shift slightly with mordant choice, fiber type, water pH, and even the season in which the plant was harvested. The same madder root mordanted with alum gives coral; mordanted with iron, it gives a deep terracotta. Weld with alum gives chartreuse; with iron, it shifts to olive. The interaction between plant chemistry and mordant chemistry creates a palette that no synthetic dye can fully replicate. 😮
Natural dyers today — working with botanical dyes and the WOF ratios that the textile guilds refined over centuries — are recovering something that the synthetic era nearly erased. The Botanical Mordant Calculator is part of that recovery. 🌿
Who Is This For? 🎯
Natural dyers working with plant-based cellulose fibers (cotton, linen, hemp) who find most guides focused on wool
Fiber artists moving from synthetic to botanical dye processes and needing precise ratios
Crafters and hobbyists working with foraged or purchased dye plants and needing exact quantities for repeatable results
Sustainable fashion enthusiasts interested in low-impact, chemical-free textile color
Textile history researchers reproducing historical dyeing processes with documented accuracy
Cellulose fiber dyeing is a significantly more involved process than protein fiber dyeing, and most beginner resources skip over the mordanting step or explain it inadequately. This tool fills that gap specifically — with WOF ratios calibrated to the same guild-derived standards that professional dyers used for centuries.
→ Try the Botanical Mordant Calculator on riatto.ovh
Free to use. No sign-up required.
🐾 Bureau of Unauthorized Fiber Investigation & Strategic Warm Bath Occupation
Field Report — Incident Reference: Warm Pot Proximity Event, Fiber Color Anomaly, Paws 1–4
i was conducting a routine kitchen patrol at approximately 11:43 when i detected elevated ambient temperature and an unusual herbal scent emanating from the stovetop area 😼
there was a pot. it was warm. this is my jurisdiction.
i approached the fiber draped over the pot edge for cooling — cotton, 150 g, marigold amber, clearly unattended and therefore available for quality assessment. i placed both front paws on it to evaluate structural integrity. 🐱 the fiber had absorbed some color. this color has now transferred to my paws. this was not my fault — the human should have secured the fiber more effectively before leaving the room. i am documenting this as a regulatory failure.
i then sat on the linen pre-mordant sample drying on the counter. it was warm and slightly damp and smelled pleasantly of tannin and sumac. this is a 10/10 sitting surface. i claimed it. 😹
the human attempted to relocate me to the floor. i returned to the linen sample within four seconds, which i believe is a personal record. i then conducted a thorough sniff-inspection of the ferrous sulfate container, which smelled alarming and suspicious. i knocked it sideways to confirm it was properly sealed. it was. i have noted “ferrous sulfate: sealed, suspicious, smells bad” in my report.
the onion-skin water bath, i now know, is not a drinking vessel. i have added this to my operational knowledge base at significant personal cost. 🙀
my paws are currently chartreuse from the weld preset. i consider this an enhancement. the human disagrees. we are at an impasse.
brrp.
— Chief Inspector Madder, Bureau of Unauthorized Fiber Investigation & Strategic Warm Bath Occupation
“The bath was warm. We had no choice.”
References
Mordant — Wikipedia (etymology from Latin mordere; aluminum coordination chemistry)
Alum — Wikipedia (history, natural deposits, mordanting applications)
Tolfa Alum Mines — Wikipedia [Flag for verification: confirm 1462 date and Papal claim]
Weld — Wikipedia (Reseda luteola, lightfast yellow)
William Henry Perkin — Wikipedia (mauveine, 1856 — first commercially mass-produced synthetic dye)
Alizarin — Wikipedia (synthetic alizarin, Graebe & Liebermann, 1869 — trigger for madder industry collapse)
The Blueprint Was Always a Photograph — riatto.substack.com (iron chemistry in cyanotype — related reading)
Before the Tube: The Oil Formula That Keeps a Painting Intact — riatto.substack.com (color chemistry companion post)
Wrapping Up
Every color in a pre-synthetic textile had to earn its permanence. The dye alone was never enough — without a mordant to bite the fiber first, it washed out. The alum trade shaped the economics of medieval cloth production for centuries, all because of one ratio: roughly 20 g of salt per 100 g of dry fiber.
The Botanical Mordant Calculator encodes that ratio — the same guild-derived standard that professional dyers used for centuries — and scales it to whatever you’re working with. A 50 g test skein or a full hemp batch, it handles the maths. The dye plant reference table gives you ten proven plant-mordant pairings. The presets load in a click.
The onion skin preset is free. It takes 30 seconds to configure. It will tell you exactly what you need, and why. 🌿
Built something with it? Have feedback? Drop it in the comments.
→ Natural dyeing mordant supplies on Amazon
Affiliate disclosure: This post contains Amazon affiliate links. I may earn a small commission at no extra cost to you.
About this article
This post was written by AI and reviewed by the author. All factual claims were verified (with another prompt) at the time of publication. Final perspective, editorial judgement, and any opinions expressed are the author’s own.Published on riatto.substack.com · March 2026